Ampa Accelerates Rollout of Portable Neuromodulation Platform for Depression with Oversubscribed Financing
PR Newswire
PALO ALTO, Calif., May 14, 2026
PALO ALTO, Calif., May 14, 2026 /PRNewswire/ -- Transcranial magnetic stimulation (TMS) has been an FDA-cleared therapy for treatment-resistant depression since 2008, but the equipment has historically been too expensive, large, and complex for most clinics to offer.
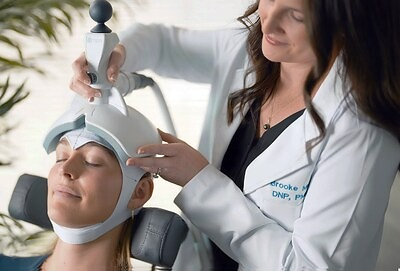
Ampa, co-founded by seminal researcher Jonathan Downar, MD, PhD, has developed a new TMS device that costs $3,000 per month, fits in the trunk of a car, and can be operated after a few hours of training.
Today, Ampa announced an oversubscribed financing that will accelerate the rollout of its FDA-cleared Ampa One TMS system, already used by clinics in more than 30 states. Ampa has also secured regulatory approval in Israel, paving the way for commercial launch overseas.
"The Ampa One device is, by far, the simplest way for clinics to add TMS to their offerings," says Don Vaughn, PhD, CEO and neuroscientist. "It can be used anywhere, by practically any licensed professional, at a fraction of the typical cost. Demand has doubled every quarter, and this most recent funding will help us meet this accelerating demand."
This most recent financing was led by a syndicate of investors including Morningside Ventures, Trimera Capital, Neta Foundation, Jake Collective, and noted technology investor and author Tim Ferriss, bringing total funding to more than $25 million.
"I've received TMS treatments myself, I've tracked the technology for almost 20 years, and Ampa is poised to offer the iPhone of the field. It's the first device I've seen that could legitimately scale to help millions of people," said Tim Ferriss. "I've personally experienced One-Day TMS treatments that have taken my symptoms from a 9 out of 10 in severity to a 1, lasting a minimum of 3-4 months. Nothing else I've tried comes close to that durability. It's a glimpse of the future of psychiatry."
"We're thrilled to partner with Ampa," said Owen Muir, MD, Co-Founder of Radial, which operates clinics nationwide that offer Spravato® (esketamine), TMS, neurofeedback, and more. "The Ampa One is a category-defining product, and we see Ampa emerging as a true platform for next-generation brain medicine therapies."
The company continues to see increasing adoption by mental health clinics who appreciate the multi-layered advantages of the Ampa One: portable hardware; a lightweight, hand-holdable coil; camera-guided targeting; pre-printed neuronavigation caps; high-throughput capacity; and affordable subscription pricing.
"Ampa is bringing innovative, life-changing tools to millions of people navigating significant mental health challenges," said Melissa Floren Filippone, CEO of Jake Collective, a nonprofit and investment engine dedicated to improving the lives of neurodivergent young people. "We are excited to be furthering Ampa's critical work with our first impact investment."
"With breakthrough innovation, Ampa is transforming the landscape and broadening access to care," added Brian Earthman, MD of Cedar Park TMS, a Beacon Behavioral Partners Clinic.
About Ampa
Ampa is a neurotechnology company creating practical tools that help people recover their mental health. The FDA-cleared Ampa One system is the most accessible, portable, easy-to-use TMS system available — built by clinicians for clinicians. Learn more and book a demonstration at www.ampahealth.com
Media Contact:
Itamar Kandel
(415) 463-3454
414551@email4pr.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/ampa-accelerates-rollout-of-portable-neuromodulation-platform-for-depression-with-oversubscribed-financing-302771815.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/ampa-accelerates-rollout-of-portable-neuromodulation-platform-for-depression-with-oversubscribed-financing-302771815.html
SOURCE Ampa